Insight into the strong aggregation-induced emission of low-conjugated racemic C6-unsubstituted tetrahydropyrimidines through crystal-structure–property relationship of polymorphs†
Abstract
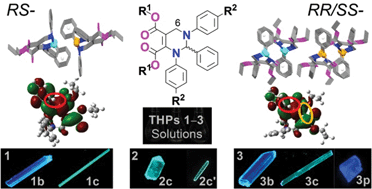
Racemic C6-unsubstituted tetrahydropyrimidines (THPs) are a series of fluorophores with a strong aggregation-induced emission (AIE) effect. However, they do not possess the structural features of conventional AIE compounds. In order to understand their AIE mechanism, here, the influences of the molecular packing mode and the conformation on the optical properties of THPs were investigated using seven crystalline polymorphs of three THPs (1–3). The racemic THPs 1–3 have low-conjugated and highly flexible molecular structures, and hence show practically no emission in different organic solvents. However, the fluorescence quantum yields of their polymorphs are up to 93%, and the maximum excitation (λex) and emission (λem) wavelengths of the polymorphs are long at 409 and 484 nm, respectively. Single-crystal structures and theoretical calculation of the HOMOs and LUMOs based on the molecular conformations of these polymorphs indicate that the polymorphs with the shortest λex and λem values possess a RS-packing mode (R- and S-enantiomers self-assemble as paired anti-parallel lines) and a more twisted conformation without through-space conjugation between the dicarboxylates, but the polymorphs with longer λex and λem values adopt a RR/SS-packing mode (R- and S-enantiomers self-assemble as unpaired zigzag lines) and a less twisted conformation with through-space conjugation between the dicarboxylates. The molecular conformations of 1–3 in all these polymorphs are stereo and more twisted than those in solution. Although 1–3 are poorly conjugated, the radiative rate constants (kr) of their polymorphs are as large as conventional fluorophores (0.41–1.03 × 108 s−1) because of improved electronic conjugation by both through-bond and through-space interactions. Based on the obtained results, it can be deduced that the strong AIE arises not only from the restriction of intramolecular motion but also from enhanced electronic coupling and radiatively-favored inter-crossed local excitation (LE) and intramolecular charge transfer (ICT) excitation states. The abnormal molecular structures, easily-controllable self-assembly of the R- and S-enantiomers, and the strong AIE effect make THPs very useful fluorophores for applications and theoretical research.


 Please wait while we load your content...
Please wait while we load your content...