Non-monotonous dependence of the electrical conductivity and chemical stability of a graphene freestanding film on the degree of reduction†
Abstract
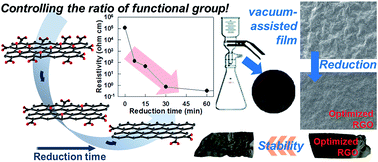
The surface morphology, electrical conductivity, and chemical stability of freestanding graphene films can be effectively tailored by controlled reduction of graphene oxide (G-O) precursor. The elastic freestanding graphene films with different degree of reduction can be fabricated by vacuum-assisted filtration of reduced graphene oxide (rG-O) nanosheets prepared with different reduction times. The surface morphology and crystal structure of the resulting rG-O films can be tuned with the change of reduction conditions. Of prime importance is that these graphene films do not show a monotonous dependence of electrical conductivity with the concentration of oxygenated functional groups. After the short reduction time of 30 min, the electrical conductivity of the rG-O films becomes saturated with retention of a considerable amount of oxygenated groups, demonstrating the rapid establishment of electron percolation paths. Interestingly, this graphene film with the reduction time of 30 min displays a higher stability with respect to microwave heating than the other films, a result of the depression of microwave absorption by the increase of electrical conductivity and the reinforced dipolar interaction between restacked graphene by the presence of oxygenated functional groups. The present work highlights the importance of controlled reduction conditions in tailoring the transport properties, surface morphology, and chemical stability of rG-O freestanding films.

 Please wait while we load your content...
Please wait while we load your content...