BF3-promoted electrochemical properties of quinoxaline in propylene carbonate†
Abstract
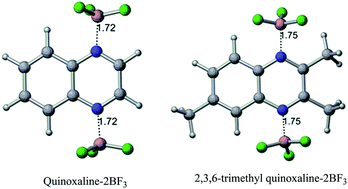
Electrochemical and density functional studies demonstrate that coordination of electrolyte constituents to quinoxalines modulates their electrochemical properties. Quinoxalines are shown to be electrochemically inactive in most electrolytes in propylene carbonate, yet the predicted reduction potential is shown to match computational estimates in acetonitrile. We find that in the presence of LiBF4 and trace water, an adduct is formed between quinoxaline and the Lewis acid BF3, which then displays electrochemical activity at 1–1.5 V higher than prior observations of quinoxaline electrochemistry in non-aqueous media. Direct synthesis and testing of a bis-BF3 quinoxaline complex further validates the assignment of the electrochemically active species, presenting up to a ∼26-fold improvement in charging capacity, demonstrating the advantages of this adduct over unmodified quinoxaline in LiBF4-based electrolyte. The use of Lewis acids to effectively “turn on” the electrochemical activity of organic molecules may lead to the development of new active material classes for energy storage applications.

 Please wait while we load your content...
Please wait while we load your content...