Nanostructured nickel sulfides: phase evolution, characterization and electrocatalytic properties for the hydrogen evolution reaction†
Abstract
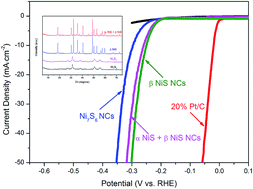
Nanostructured nickel sulfide with different phases was synthesized via a thermal decomposition approach using nickel acetylacetonate as the nickel source, 1-dodecanethiol as the sulfur source and oleylamine as the high boiling solvent. The phase evolution of nickel sulfide nanocrystals (NCs) can be easily achieved by changing the molar ratio of Ni : S precursor and the species of the solvent. X-ray diffraction (XRD), transmission electron microscopy (TEM), scanning electron microscopy (SEM), energy dispersive spectroscopy (EDS) mapping, X-ray photoelectron spectroscopy (XPS) and N2 adsorption–desorption were used to characterize the as-synthesized nickel sulfide NCs. The electrocatalytic activity and stability of the as-synthesized nickel sulfide NCs for the hydrogen evolution reaction (HER) were investigated. Due to the different crystalline phase structures of the as-synthesized nickel sulfide NCs, the β NiS NCs exhibit better electrocatalytic activity with a low onset overpotential (186 mV), a small Tafel slope (51.2 mV dec−1), a high exchange current density (2.4 × 10−6 A cm−2), a large electrochemical double-layer capacitance (2.7 mF cm−2) and improved stability than the Ni7S6 and α NiS + β NiS NCs for HER. This study provides a good strategy for designing more efficient nickel sulfide catalysts for the HER.

 Please wait while we load your content...
Please wait while we load your content...