Controllable dye adsorption behavior on amorphous tungsten oxide nanosheet surfaces†
Abstract
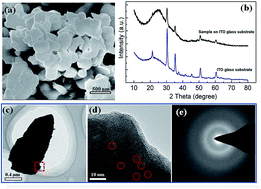
In this study, the surface interactions between different dyes and amorphous WO3 nanosheets in water were investigated. The amorphous WO3 nanosheets had a strong adsorption for cationic dyes and could be self-precipitated from water after enough dye molecules were absorbed into their micro-structures. The absorbed cationic dyes eventually formed H-aggregates or J-aggregates on the surface of WO3. In contrast, the amorphous WO3 nanosheets had a weak or no adsorption capacity for anionic dyes. This selective adsorption property of WO3 nanosheets could be applied to separate cationic dyes from a mixed solution. The possibilities of pH-induced desorption for cationic dyes on the WO3 nanosheet surfaces have also been demonstrated in this study, and its mechanism has been discussed in detail. The findings leaded to a better understanding of the dye adsorption behavior on amorphous tungsten oxide surfaces.

 Please wait while we load your content...
Please wait while we load your content...