Tunable ternary (P, S, N)-doped graphene as an efficient electrocatalyst for oxygen reduction reaction in an alkaline medium†
Abstract
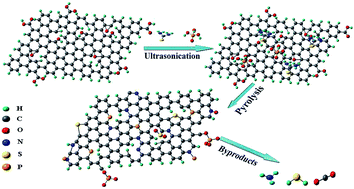
In this work, we utilize a one-step pyrolysis method to thermally synthesize phosphorus (P), sulfur (S) and nitrogen (N) ternary-doped graphene (PSNG) using graphene oxide (GO), phosphoric acid and thiourea precursors as a low-cost and highly-efficient catalyst for the oxygen reduction reaction (ORR). The synthesized PSNG was characterized by transmission electron microscopy (TEM), X-ray diffraction (XRD), Raman spectroscopy, and X-ray photoelectron spectroscopy (XPS). The electrocatalytic activity of the PSNG composite towards ORR was evaluated by the linear sweep voltammetry (LSV) method. Electrochemical measurements reveal that the pyrolyzed PSNG at 1 : 10 mass ratio of phosphoric acid to thiourea has excellent catalytic activity towards ORR in an alkaline electrolyte, including large kinetic-limiting current density and good stability as well as a desirable four-electron pathway for the formation of water. These superior properties make the PSNG a promising cathode catalyst for alkaline fuel cells.

 Please wait while we load your content...
Please wait while we load your content...