Self-microemulsifying drug delivery system of curcumin with enhanced solubility and bioavailability using a new semi-synthetic bicephalous heterolipid: in vitro and in vivo evaluation†
Abstract
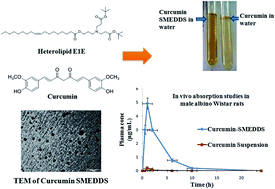
Despite the various therapeutic actions displayed by curcumin, its poor aqueous solubility and bioavailability restricts its biological applications. This problem can be addressed by its efficient loading into a nano formulation. The present paper reports the utilization of a new semi-synthetic oleic acid derived bicephalous heterolipid, E1E, as an oil phase in the formulation development of a self-microemulsifying drug delivery system (SMEDDS) of curcumin to enhance its solubility and bioavailability. The solubility of curcumin in E1E was found to be 14 and 2.6-fold greater than oleic acid and ethyl oleate respectively. The SMEDDS developed from E1E (Curcumin-E1E_SMEDDS5e) had high curcumin loading efficiency of 70.52 ± 2.46 mg g−1, and was able to form spontaneous microemulsion on addition to aqueous phase with mean globule diameter of 22.39 ± 0.2 nm and polydispersity index of 0.243 ± 0.010. In vivo oral bioavailability studies in male Wistar rats revealed that the maximum serum concentration (Cmax) and time taken to reach maximum serum concentration (Tmax) were 4.921 ± 0.42 μg mL−1 and 60 min respectively. The absorption of curcumin increased 26-folds via its delivery through Curcumin-E1E_SMEDDS5e. The cytostatic and total growth inhibition concentrations of Curcumin-E1E_SMEDDS5e against HeLa cell line were almost comparable to doxorubicin (GI50 and TGI < 10 μg mL−1) whereas lethal concentration (LC50) was 71.8 μg mL−1. Further, the minimum inhibitory concentrations against E. coli and S. aureus were 48.62 and 97.65 μg mL−1 respectively. These results suggested potent anticancer, antibacterial activity and non-cytotoxicity of developed Curcumin-E1E_SMEDDS5e. The heterolipid, E1E, proved to be an efficient solubility and bioavailability enhancer for curcumin for formulation development of its SMEDDS.

 Please wait while we load your content...
Please wait while we load your content...