Competitive lithium and sodium intercalation into sodium manganese phospho-olivine NaMnPO4 covered with carbon black
Abstract
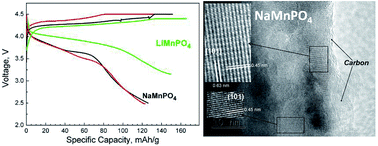
In this contribution we provide novel data on the reversible lithium and sodium ion intercalation into a sodium-manganese phospho-olivine NaMnPO4, when it is used as a cathode in model lithium-ion cells. The ion-exchange reaction involving the participation of KMnPO4·H2O dittmarite as precursor was chosen for the preparation of NaMnPO4. The NaMnPO4 particles were covered with carbonaceous materials to improve the electrical conductivity and electrolyte wetting. The procedure includes ball-milling of NaMnPO4 with conductive carbon black additives Super C/65, followed by thermal treatment. The mechanically treated samples consist of well crystallized phospho-olivine phase NaMnPO4 free of any anti-site defects and disordered carbon species with graphite like medium-range order. The composite NaMnPO4/C material manifests a reversible capacity between 80–85 mA h g−1 in model lithium cells versus lithium anode. Prior to the electrochemical test, the chemical inertness of NaMnPO4 in the lithium electrolyte is studied by soaking phospho-olivines in the solution of LiPF6 in EC:DMC. The mechanism of the reversible intercalation/deintercalation cycling is investigated using ex situ X-ray powder diffraction, TEM and high-angle annular dark field STEM analysis, infrared spectroscopy and electron paramagnetic resonance spectroscopy (EPR). The study demonstrates, for the first time, that NaMnPO4 is able to intercalate reversibly both Na+ and Li+ ions following the chemical reaction LixNa1−xMnPO4 ↔ Li0.0Na0.5MnPO4 (0.25 ≤ x ≤ 0.45).

 Please wait while we load your content...
Please wait while we load your content...