Extraction of preformed graphene oxide from coal: its clenched fist form entrapping large molecules†
Abstract
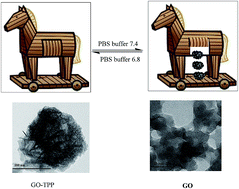
Graphene oxide, (GO), present in low grade coal, formed under the natural coalification of plants is extracted by leaching with cold nitric acid. A similar easy extraction of GO is followed for wood charcoal. Such GO sheets are thermally not very stable and on aging are fragmented into small spherical shapes. These can be solubilized in 10% NaOH and reprecipitated by adjusting the pH to around 7. Further purification could be achieved by extracting with ethanol as these shapes are highly soluble. This GO is larger than a graphene quantum dot and can be folded into a closed fist form and it is shown to entrap tetraphenylporphyrin (TPP). The TPP in the composite is not soluble in dichloromethane whereas free TPP is highly soluble. Such a GO–TPP composite retains the properties of pure GO as it is soluble in ethanol in which free TPP is insoluble. It is shown that in phosphate buffered saline (PBS, pH, 6.8) this composite is soluble and remained intact. At a higher pH of PBS buffer it releases all entrapped TPP by opening the clenched fist form. Similarly the drug donepezil, used in Alzheimer’s treatment gets entrapped by GO which is also released at a higher pH, 7.4. The clenched fist form of corrugated GO is related to the pH dependent epoxide dominated structure as proposed by Hofmann and Holst’s model and the open structure may be dominated by hydroxyl groups (enol) following Ruess’s model. Such pH dependent inter-conversion uniquely enables GO to act as a Trojan horse in carrying and releasing a drug molecule under a physio-compatible environment.

 Please wait while we load your content...
Please wait while we load your content...