Surface immobilization of β-cyclodextrin on hybrid silica and its fast adsorption performance of p-nitrophenol from the aqueous phase
Abstract
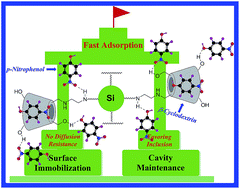
Renewable β-cyclodextrin (β-CD) was immobilized onto the surface of hybrid silica using ethylenediamine as linking groups to construct an adsorbent in water treatment (CD@Si), and the obtained CD@Si was characterized through FT-IR, XPS, EDX, contact angle measurement, TGA, solid-state 13C NMR, SEM, and XRD analyses. The effect of initial pH, contact time on the adsorption performance of CD@Si for p-nitrophenol, and the adsorption kinetics, adsorption isotherms, adsorption thermodynamics, reusability and adsorption mechanism were investigated systematically, which indicate that the adsorption of p-nitrophenol onto CD@Si is a very fast process. The adsorption equilibrium can be reached in 15 s with an acceptable equilibrium adsorption capacity of 69.6 mg g−1 at pH 7.0, which is much faster than many reported adsorbents based on β-CD. The adsorption of p-nitrophenol onto CD@Si follows the pseudo-second-order model, obeys the Freundlich model, and is a feasible, spontaneous, and exothermic process which is more favorable at lower temperatures. And the formation of an inclusion complex and a hydrogen bond interaction are two origins of p-nitrophenol being adsorbed onto CD@Si. Additionally, CD@Si can be recycled and reused for at least five runs with an acceptable adsorption capacity, and is a very promising adsorbent for the fast adsorption of p-nitrophenol or its analogues from the aqueous phase. Additionally, this work also provides a strategy to increase the adsorption rate of adsorbents based on β-CD.

 Please wait while we load your content...
Please wait while we load your content...