Density functional studies of zirconia with different crystal phases for oxygen reduction reaction
Abstract
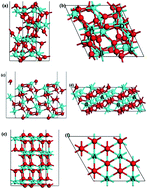
To understand the origin of its catalytic activity, the oxygen reduction reaction (ORR) on zirconia with different phases is investigated by the first principles method. The free energy diagrams for an associative mechanism are firstly given to identify the rate-determining step for the ORR on the (111) surface of monoclinic, tetragonal and cubic phase zirconia. The calculation results show that the first electron transfer is the rate-determining step for the ORR on monoclinic and cubic phase zirconia, whereas the last electron transfer is the rate-limiting step for the ORR on tetragonal phase zirconia. The d-electron partial density of states for the active zirconium atoms is calculated to explore the relationship between the ORR catalytic activity of zirconia with different phases and their electronic properties. The calculated results show that the catalytic activity of zirconia for ORR is strongly dependent on the d-orbital occupation of the active zirconium atoms.

 Please wait while we load your content...
Please wait while we load your content...