Influence of electron acceptors on the kinetics of metoprolol photocatalytic degradation in TiO2 suspension. A combined experimental and theoretical study†
Abstract
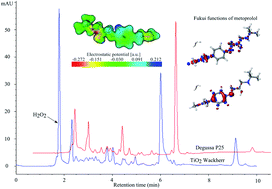
Metoprolol (MET) belongs to a group of frequently used β1-blockers, which often occur in waste waters. The objective of this work was to employ liquid chromatography (LC) and total organic carbon methods to study the photocatalytic degradation of MET in UV irradiated aqueous suspensions of TiO2 (Wackherr's “Oxyde de titane standard” and Degussa P25), in the presence of different electron acceptors such as molecular oxygen, hydrogen peroxide, potassium bromate, and ammonium persulfate. The degradation rates were found to be strongly influenced by the kind of electron acceptor and the type of catalyst. The optimal amount of hydrogen peroxide and potassium bromate was investigated as well. MET photocatalytic degradation was the fastest in the presence of O2 and potassium bromate with TiO2 Degussa P25, while mineralization was most efficient in the presence of molecular oxygen alone. In all investigated cases, degradation followed a pseudo-first order kinetics. Reaction intermediates of MET degradation in the presence of different electron acceptors with both catalysts were studied in detail and a number of them were indentified using LC-ESI-MS/MS. The interactions with MET of reactive radical species relevant to this study (O2˙−, ˙OH, BrO2˙, and SO4˙−) were theoretically investigated by means of density functional theory (DFT) computations.

 Please wait while we load your content...
Please wait while we load your content...