Di-tert-butyl peroxide (DTBP) promoted dehydrogenative coupling: an expedient and metal-free synthesis of oxindoles via intramolecular C(sp2)–H and C(sp3)–H bond activation†
Abstract
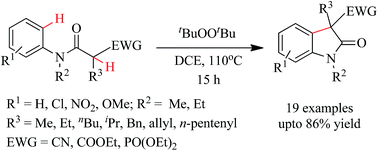
An efficient di-tert-butyl peroxide (DTBP) promoted synthesis of oxindole has been developed. This methodology involves C(sp3)–H and C(sp2)–H bond activation under metal-free conditions. This synthetic approach towards oxindole synthesis avoids bases and hazardous iodine reagents unlike other methodologies developed so far. This metal- and base-free protocol is operationally simple and ecofriendly. It provides an expedient approach to access oxindoles in moderate to very good yields in DCE at 110 °C.

 Please wait while we load your content...
Please wait while we load your content...