Liposome-based gene delivery systems containing a steroid derivative: computational and small angle X-ray diffraction study†
Abstract
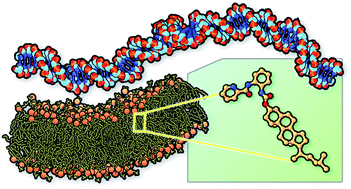
In this study, the structural properties and the phase behaviour of mixed composition neutral liposomes containing a functionalized steroid are reported. With the aim to design neutral liposomes able to coordinate cations and to complex DNA, we synthesized cholesteryl-2-(picolinamido)-phenylcarbamate (CHOLp) containing an N-aryl picolinamide group as chelating agent linked to the steroid structure via a carbamate moiety. The phase behaviour of mixtures of the functionalized cholesterol (CHOLp) and dioleoyl-phosphatidylcholine (DOPC) was investigated by means of X-ray diffraction. Simultaneously, atomistic molecular dynamics simulations of DOPC/CHOLp bilayers as a function of CHOLp molar fractions were carried out to investigate the specific effects of the polar steroid on the structural and dynamic properties of these zwitterionic bilayers. The molecular modelling studies have been performed both in absence and in presence of bivalent cations salts in order to assess the CHOLp ability to coordinate metal ions. The results show good stability of the resulting DOPC/CHOLp bilayers which is improved by the presence of salt. This is particularly evident at low amount of CHOLp where a high order of the lipid tails can be observed, suggesting stabilization of the corresponding DOPC liposomes. This feature can be ascribed to the polar nature and structural properties of the ligand. In fact, due to the presence of the aromatic moieties, CHOLp combines two different behaviours, namely a propensity to realize both intermolecular π-stacking interactions and cation–π bonding mainly evident in CaCl2. The last feature confirms for CHOLp a role as a cation-mediated complexation agent for DNA. The X-ray diffraction data on the capability of DOPC/CHOLp liposomes to complex DNA was also reported.

 Please wait while we load your content...
Please wait while we load your content...