First noscapine glycoconjugates inspired by click chemistry†
Abstract
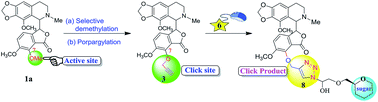
A number of novel 7-O-noscapine glycoconjugates have been synthesized starting from noscapine, an alkaloid found in the opium plant, via two successive steps. The first step is a selective 7-O-demethylation of noscapine and the next is a subsequent propargylation which affords 7-O-propargyl noscapine (3) in good yield. The structure was confirmed by extensive spectroscopic data including single crystal X-ray data. The 1,3-dipolar cycloaddition of the developed noscapine derivative 3 with glycosyl azides 6a–m was investigated to give the triazole-linked second-generation noscapine analogs in their glycoconjugate forms (8a–m) to augment the therapeutic efficacy of noscapine.

 Please wait while we load your content...
Please wait while we load your content...