What causes tumbling of altro-α-CD derivatives? Insight from computer simulations†
Abstract
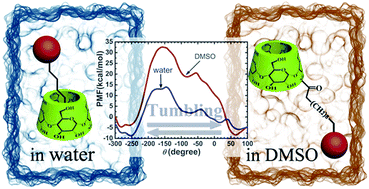
In water, a remarkable motion can be observed with a [2]rotaxane, wherein the rotor translocates by reeling its axle in the cavity of an altro-α-CD stopper. Similarly, in aqueous solution, an alkyl altro-α-CD dimer reels its alkyl chain in the altro-α-CD cavity to form a pseudo[1]rotaxane dimer. This reeling motion is in fact induced by the tumbling of the altropyranose unit of an altro-α-CD, a process shown to be solvent-dependent. Tumbling, however, does not occur in low-polarity solvents such as methanol and DMSO. In the present contribution, the mechanism that underlies solvent-controlled tumbling has been studied at the atomic level by means of molecular dynamics simulations combined with microsecond-timescale free-energy calculations. The free-energy profile delineating the tumbling in water of the altropyranose unit of an alkyl altro-α-CD indicates that a 19.8 kcal mol−1 barrier must be overcome to yield the self-inclusion complex, which is the most stable state available to the supramolecular assembly. In DMSO, the free-energy barrier is about 21.0 kcal mol−1 higher, and the self-included alkyl altro-α-CD corresponds to a metastable state. These results provide new thermodynamic and kinetic insights into solvent-controlled tumbling, and reveal the essence of different experimental observations. Further investigation shows that aside from the polarity of the solvent, tumbling of the altro-α-CD derivative stems from the hydrophobicity of the side chain and the propensity of the former to include the latter, which opens perspectives for the design of new, related supramolecular assemblies.

 Please wait while we load your content...
Please wait while we load your content...