Organogel formation via supramolecular assembly of oleic acid and sodium oleate
Abstract
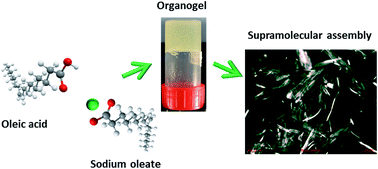
To create materials with novel functionalities, the formation of gels within hydrophobic media has become popular. This is often accomplished through the assembly of low molecular weight organogelators into a variety of complex phases through intermolecular interactions. In the case of edible materials, the assembly of saturated fatty acids to form fat crystal networks is often used for structuring. Here, the first example of structuring with unsaturated fatty acids is reported, namely mixtures of oleic acid and sodium oleate, to structure edible lipid phases. Small-angle scattering demonstrates that the resultant structures, which vary with oleic acid and sodium oleate molar ratio, comprise either inverse micellar or lamellar phases, combined with the formation of crystalline space-filling networks. Network formation was found for filler concentrations above 10 wt%. Rheological measurements show that gel strength depends on the ratio of oleic acid to sodium oleate, and is greater when only oleic acid is used. The addition of up to 1.5 wt% of water enhanced the strength of the organogels, probably through supplementary hydrogen bonding but, for concentrations greater than 2.0 wt%, the assembly was inhibited leading to collapse of the gel.

 Please wait while we load your content...
Please wait while we load your content...