Genetic incorporation of d-amino acids into green fluorescent protein based on polysubstrate specificity†
Abstract
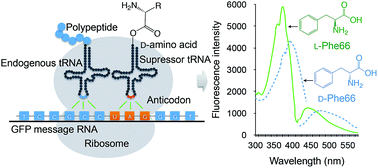
D-amino acids are widely distributed in living organisms and are suggested to play important roles in protein folding and function. Genetic incorporation of D-amino acids through protein synthesis machinery has been an exploratory task in protein engineering, and is greatly enticing but difficult. In the present work, a number of D-amino acids were genetically incorporated into green fluorescent protein using a polysubstrate-specific tRNA synthetase with cognate tRNA in Escherichia coli, and the GFPuv mutant containing D-phenylalanine in the fluorophore at residue 66 was characterized. Stereochemical switching of phenylalanine at position 66 resulted in red shifts in the emission and excitation maxima and significantly improved the thermal stability of the protein. Molecular modeling further revealed that the opposite configurations of Phe66 in GFPuv produced two respective isomeric fluorophores that exhibit distinctive spectral properties and thermal stability. The present study expands the backbone stereochemistry of protein molecules by in vivo ribosomal translation to facilitate protein engineering.

 Please wait while we load your content...
Please wait while we load your content...