The enantiopreference in the solid state probed in lamivudine crystal forms with mandelic acid†
Abstract
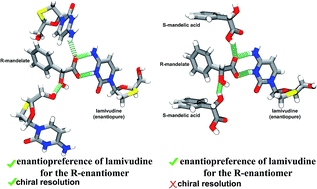
The separation of enantiomers from a racemic mixture, known as chiral resolution, has been achieved nowadays through the preparation of multicomponent molecular crystals. Here we have investigated the crystallization of lamivudine, an enantiopure anti-HIV/HBV drug, with the enantiomers of mandelic acid. First, chiral resolution has been obtained here through salt formation. Lamivudine R-mandelate was isolated from an isopropyl alcohol solution of the drug and racemic mandelic acid in 1 : 2 stoichiometry. The robust two-point 2-aminopyridinium-carboxylate synthon was identified in this structure. Second, in the course of our investigations to know if diastereomeric salts could be formed from other solvent environment, S-mandelic acid cocrystal of lamivudine R-mandelate trihydrate has grown from a solution of water and ethyl alcohol loading the drug and racemic mandelic acid (lamivudine : racemic mandelic acid 1 : 2 stoichiometry). It is an example of a cocrystal of a salt whose enantiopreference was maintained for ionic pair formation between a lamivudinium cation and R-mandelate anion. Although chiral resolution was not viable in this structure, the robust synthon present in the salt phase was conserved even if both mandelic acid enantiomers are present together in the crystal lattice. Therefore, we here demonstrated the enantiopreference in the solid state even if chiral resolution does not occur.

 Please wait while we load your content...
Please wait while we load your content...