Magnesium carbonate basic coating on cotton cloth as a novel adsorbent for the removal of uranium†
Abstract
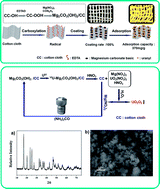
The magnesium carbonate basic coating on the cotton cloth (Mg2CO3(OH)2/CC) was prepared by a facile and cost-effective method for uranium(VI) adsorption. The process of the obtained material for uranium(VI) adsorption from an aqueous solution was fully researched. The results reveal that the maximum adsorption capacity of the Mg2CO3(OH)2/CC toward uranium is 370 mg g−1, providing a strong efficiency for the removal of uranium from aqueous solution. The experimental data were analyzed using adsorption kinetic models. The adsorption kinetics of uranium onto Mg2CO3(OH)2/CC obey the pseudo-second-order model, indicating that the determining step might be chemical adsorption. The calculated thermodynamic parameters, such as ΔHo, ΔSo and ΔGo, show that the adsorption process is spontaneous and endothermic. Furthermore, the as-prepared material can be easily separated from the aqueous solution after adsorption. Such excellent performance may promote this promising and effective adsorbent for practical uranium(VI) adsorption.

 Please wait while we load your content...
Please wait while we load your content...