Theoretical studies of a 3D-to-planar structural transition in SinAl5−n+1,0,−1 (n = 0–5) clusters†
Abstract
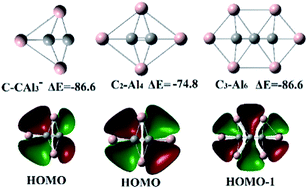
Basin-hopping global searching and quantum chemistry calculations were performed to predict global minimum structures of SinAl5−n+1,0,−1 (n = 0–5) clusters in order to explore the intrinsic mechanism of 3D-to-planar structural transition and structural characteristics of planar tetracoordinate species. A structural similarity of isoelectronic SinAl5−n+1,0,−1 (n = 0–5) clusters exists, and may generally be extended to similar clusters containing B, C, N, O, and P. The structural diagram and molecular orbital analysis reveal that a 3D-to-planar structural transition should be related to composition and total valence electron number of clusters. Our calculations indicate that the global minima of SinAl5−n+1,0,−1 (n = 0–5) with planar tetracoordinate Si (ptSi) should meet the 18-electron rule and take the charge of −1, 0, +1. Further, it is found that 18-electron M–Al4 (M = B, C, N, O, and F) planar clusters prefer a central atom M with a low electronegativity and peripheral Al substituted with a lower-electronegativity atom. Based on the structural characteristics of planar clusters, we firstly predict a novel planar tetracoordinate C (PtC) structure C2Al3− which is more stable in energy than the experimentally observed CAl42−. C2Al3− may become a building block to assemble some larger supermolecule containing multiple planar hypercoordinate C (phC).

 Please wait while we load your content...
Please wait while we load your content...