Mesoporous ZnCo2O4 microspheres as an anode material for high-performance secondary lithium ion batteries
Abstract
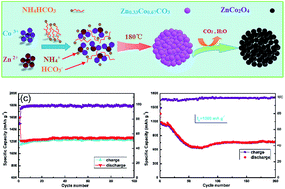
Herein, we report mesoporous ZnCo2O4 microspheres fabricated by a facile hydrothermal method followed by pyrolysis of a Zn0.33Co0.67CO3 precursor. The obtained ZnCo2O4 microspheres were made up of closely packed primary nanoparticles with a diameter of about 30 nm and a large number of pores that were sized between 10 to 40 nm, which results in a high BET surface area of 39.52 m2 g−1. The large surface area permits a high interfacial contact area with the electrolyte and provides more locations and channels for fast Li+ insertion/extraction into the electrode material. The porous structure may not only be beneficial for Li+ ions to diffuse efficiently to active material with less resistance but also to buffer the volume expansion during the discharging/charging processes. When used as an anode material, the specific capacity was maintained at a high value of 1256 mA h g−1 after 100 cycles at a current density of 100 mA g−1, which is about 3.4 times larger than that of the commercial graphite electrode (372 mA h g−1). More interestingly, a reversible capacity as high as 774 mA h g−1 could be retained at a high current density of 1000 mA g−1 after 200 cycles, which indicates that the mesoporous ZnCo2O4 microspheres had excellent cycling performance at a high current density for use as anode materials for lithium-ion batteries (LIBs).

 Please wait while we load your content...
Please wait while we load your content...