Self-supported composites of thin Pt–Sn crosslinked nanowires for the highly chemoselective hydrogenation of cinnamaldehyde under ambient conditions†
Abstract
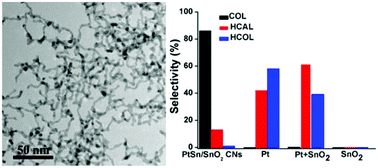
Alloying a second metal in a parent metal is an efficient way to improve the catalytic performances of monometallic nanocatalysts, thus the understanding of the alloying effects on the catalytic properties of bimetallic nanocatalysts is of great significance. Herein, we demonstrate the synthesis of self-supported composites of thin (ca. 3.8 nm) Pt–Sn crosslinked nanowires (PtSn/SnO2 CNs) with a Pt : Sn molar ratio of 3–4 : 1 via a one-pot hydrothermal method. The growth mechanism of the CNs consisted of spontaneous hydrolysis and reduction of metal ions in water solution and the subsequent oriented attachment of M(OH)x (M = Pt, Sn) particulate species under reduction by poly(vinylpyrrolidone) during hydrothermal processing. The nature of metal precursors, capping reagents and reduction rate has shown great influences on the shaping of the nanocatalysts into CNs. The PtSn/SnO2 catalyst exhibited 87% selectivity (superior over pure Pt nanoparticles (NPs) and physical mixtures of Pt and SnO2 NPs) under ambient conditions for the hydrogenation of cinnamaldehyde, owing to the formation of a Pt–Sn alloy in the CNs. The PtSn/SnO2 catalyst also showed good recyclability and stability in chemoselectivity for cycle experiments of the catalysts.
- This article is part of the themed collection: Celebrating 110th Anniversary of Chemistry at Peking University

 Please wait while we load your content...
Please wait while we load your content...