Macroscopic polarity control with alkali metal cation size and coordination environment in a series of tin iodates†
Abstract
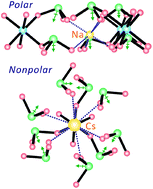
A series of stoichiometrically similar tin(IV) iodates, A2Sn(IO3)6 (A = Li, Na, K, Rb, Cs) and Sn2+Sn4+(IO3)6 have been hydrothermally synthesized. X-ray diffraction was used to determine the crystal structures of the reported materials. All six materials reveal zero-dimensional molecular structures that consist of SnO6 octahedra and IO3 polyhedra. However, the size and coordination environment of cations significantly influence the macroscopic centricities of the materials. While Li2Sn(IO3)6 and Na2Sn(IO3)6 crystallize in the noncentrosymmetric (NCS) polar space group, P63, the K, Rb, Cs, and Sn phases crystallize in the centrosymmetric (CS) nonpolar space group, R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) . Infrared and UV-vis spectroscopy, thermal analyses, and local dipole moment calculations are reported. With NCS polar materials, powder second-harmonic generation (SHG) properties, polarization, and piezoelectric measurements are also presented. The NCS properties are mainly attributable to the parallel alignment of the lone pairs in I5+ cations.
. Infrared and UV-vis spectroscopy, thermal analyses, and local dipole moment calculations are reported. With NCS polar materials, powder second-harmonic generation (SHG) properties, polarization, and piezoelectric measurements are also presented. The NCS properties are mainly attributable to the parallel alignment of the lone pairs in I5+ cations.
- This article is part of the themed collections: HOT articles in Inorganic Chemistry Frontiers for 2015 and Crystal engineering for molecular materials

 Please wait while we load your content...
Please wait while we load your content...