Doubly-responsive hyperbranched polymers and core-crosslinked star polymers with tunable reversibility†
Abstract
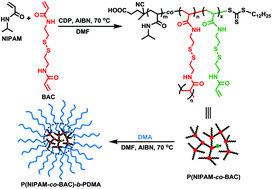
Thermo- and redox-responsive hyperbranched copolymers were prepared by statistical copolymerization of N-isopropylacrylamide (NIPAM) and N,N′-bis(acryloyl)cystamine (BAC) by reversible addition–fragmentation chain transfer (RAFT) polymerization. Kinetic studies revealed that the molecular weight of the resulting poly(NIPAM-co-BAC) gradually increased during the polymerization, and control over molecular weight and degree of branching was demonstrated. The hyperbranched copolymers showed thermoresponsive self-assembly, as determined by dynamic light scattering and turbidity measurements. Hyperbranched poly(NIPAM-co-BAC) copolymers were further used for the synthesis of star copolymers by chain extension with N,N-dimethylacrylamide. The resulting star copolymers with hyperbranched cores and linear arms were readily degraded under reducing conditions due to the divinyl crosslinker containing a redox-sensitive disulfide linkage. Not only did the disulfide bonds result in macromolecules with redox-responsive behavior, but the ability to cleave and characterize the individual branches of the hyperbranch polymer after synthesis allowed us to confirm the controlled nature of RAFT polymerization in the presence of divinyl compounds.

 Please wait while we load your content...
Please wait while we load your content...