One-pot synthesis of hyperstar polymers via sequential ATRP of inimers and functional monomers in aqueous dispersed media†
Abstract
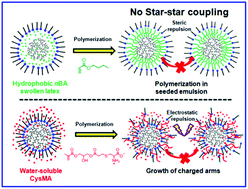
Core–shell structured hyperstar polymers that contained a hyperbranched core and hundreds of radiating arms were synthesized in a one-pot process without worrying about the hyperstar–hyperstar coupling reactions. The synthesis started with the atom transfer radical polymerization of an inimer in a microemulsion to produce hyperbranched polymers with high molecular weight, low polydispersity, and a high density of bromine initiating groups. After the complete conversion of the inimer, a second batch of monovinyl monomers was added in situ without destabilizing the microemulsion to subsequently grow radiating arms from these hyperbranched polymer macroinitiators (MIs). Two scenarios of arm growth were presented to demonstrate the efficient synthesis of hyperstar polymers. The hydrophobic n-butyl acrylate (nBA) monomer diffused into the latexes and swelled the hyperbranched polymers to form a seeded emulsion, which protected the growth of PnBA arms from each individual hyperbranched MI with no hyperstar–hyperstar coupling product even at >90% nBA conversion. The use of the zwitterionic cysteine methacrylate (CysMA) monomer caused the growth of arms out of the micelles and stabilized the hyperstar polymers in the aqueous phase, benefiting from the electrostatic repulsion between the charged arms and stars, which also avoided the hyperstar–hyperstar coupling at high conversion. When inimers containing a disulfide linker group were used in the synthesis of hyperbranched MIs, the produced hyperstar polymers exhibited rapid core degradation in a reducing environment and produced linear polymers as the degradation product.

 Please wait while we load your content...
Please wait while we load your content...