Heteroatom substituted naphthodithiophene–benzothiadiazole copolymers and their effects on photovoltaic and charge mobility properties†
Abstract
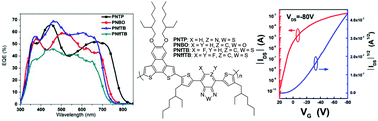
A novel series of heteroatom-substituted naphthodithiophene and dithienylbenzothiadiazole alternating copolymers namely PNTP, PNBO, PNfTB, and PNffTB were designed and synthesized in which heteroatoms including fluorine, oxygen or nitrogen were incorporated into electron-deficient benzothiadiazole units of the resulting copolymers. In general, with the incorporation of heteroatoms into the acceptor units of the polymer backbone, the absorption spectra would shift to a longer wavelength, indicating a decrease in the optical band gap. In addition, fluorine substitution can effectively lower both the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) energy levels of the resulting copolymers; on the other hand, the incorporation of nitrogen or oxygen atoms into the copolymer backbone can only stabilize the LUMO energy level of copolymers. The effect of heteroatoms on the photovoltaic and charge mobility properties of organic solar cells (OSCs) and organic field-effect transistors (OFETs) was also investigated. The bulk heterojunction (BHJ) OSCs fabricated from the blend of heteroatom-substituted copolymers and PC71BM using diiodooctane as a solvent additive with ZnO/Al as a double interlayer in an inverted device structure afforded a power conversion efficiency up to 5.05%. In addition, the solution-processed bottom gate bottom contact based OFETs fabricated from PNfTB exhibited an enhanced hole mobility of 0.14 cm2 V−1 s−1 with an on/off current ratio of 107. Our results suggested that incorporation of heteroatoms into the donor–acceptor polymer backbone would be a useful strategy to enhance the functional properties for applications in OSCs and OFETs.

 Please wait while we load your content...
Please wait while we load your content...