A new design of ionic complexation and its application for efficient protection of proteins†
Abstract
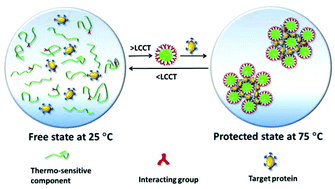
Ionic complexation is one of the most important topics in the fields of biology, physics, chemistry, and materials science. An ionic complex normally has an upper critical complexation temperature (UCCT), i.e. the ionic complex disappears above UCCT. Herein we have for the first time demonstrated that a new ionic complex, in contrast to the UCCT complex, has a lower critical complexation temperature (LCCT), which means that the ionic complex exists above UCCT but disappears below LCCT. We have further shown that the LCCT ionic complexation can efficiently protect proteins at the denaturation temperature but automatically release proteins at room temperature to freely interact with the substrates. For example, 70–80% enzymatic activity was retained after heating at 70–75 °C for 60–90 min and cooling to room temperature using this strategy. Thus this new LCCT ionic complexation would provide a cost-effective approach to protecting proteins for various biomedical applications.

 Please wait while we load your content...
Please wait while we load your content...