A molecular insight into the phototoxic reactions observed with vemurafenib, a first-line drug against metastatic melanoma
Abstract
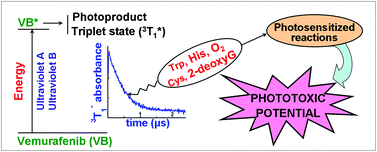
The electronic properties of vemurafenib (VB) provide a rational basis for understanding its strong UVA-induced phototoxicity. Thus, solvation of hydrophobic VB by hydrogen bonding solvents controls its photophysical, photochemical and photosensitizing properties. Addition of phosphate buffered saline (PBS) to methanol (MeOH) induces a bathochromic shift of the VB absorbance spectrum and a fluorescence emission (λmax = 450 nm, quantum yield (Φ) = 0.011). Phosphorescence (λmax = 461 nm) is observed at 77 K in MeOH. 308 nm laser flash spectroscopy demonstrates that the lifetimes (τ) and quantum yields of the VB triplet state 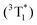 in deaerated MeOH (τMeOH = 0.41 μs, λmax ∼ 380 nm), MeOH–PBS and HSA solutions markedly depend on the microenvironment. A long-lived radical (half-life >200 μs) is also formed. The
in deaerated MeOH (τMeOH = 0.41 μs, λmax ∼ 380 nm), MeOH–PBS and HSA solutions markedly depend on the microenvironment. A long-lived radical (half-life >200 μs) is also formed. The 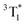 state is quenched by O2 and electron donors (Cys and 2′-deoxyguanosine) at a rate constant >1 × 109 M−1 s−1. UVA-irradiation of VB in air-saturated MeOH or MeOH–PBS solutions produces a UVA-absorbing photoproduct (Φ ∼ 5 × 10−4). VB photosensitizes Trp destruction by type I (radical formation) and type II (singlet oxygen (1O2) formation) photodynamic reactions (Φ = 0.005). Singlet oxygen production is further demonstrated by the VB-photosensitized His oxidation (ΦMeOH = 0.006).
state is quenched by O2 and electron donors (Cys and 2′-deoxyguanosine) at a rate constant >1 × 109 M−1 s−1. UVA-irradiation of VB in air-saturated MeOH or MeOH–PBS solutions produces a UVA-absorbing photoproduct (Φ ∼ 5 × 10−4). VB photosensitizes Trp destruction by type I (radical formation) and type II (singlet oxygen (1O2) formation) photodynamic reactions (Φ = 0.005). Singlet oxygen production is further demonstrated by the VB-photosensitized His oxidation (ΦMeOH = 0.006).

 Please wait while we load your content...
Please wait while we load your content...