A few key residues determine the high redox potential shift in azurin mutants†
Abstract
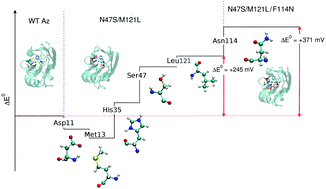
The wide range of variability of the reduction potential (E0) of blue-copper proteins has been the subject of a large number of studies in the past several years. In particular, a series of azurin mutants have been recently rationally designed tuning E0 over a very broad range (700 mV) without significantly altering the redox-active site [Marshall et al., Nature, 2009, 462, 113]. This clearly suggests that interactions outside the primary coordination sphere are relevant to determine E0 in cupredoxins. However, the molecular determinants of the redox potential variability are still undisclosed. Here, by means of atomistic molecular dynamics simulations and hybrid quantum/classical calculations, the mechanisms that determine the E0 shift of two azurin mutants with high potential shifts are unravelled. The reduction potentials of native azurin and of the mutants are calculated obtaining results in good agreement with the experiments. The analysis of the simulations reveals that only a small number of residues (including non-mutated ones) are relevant in determining the experimentally observed E0 variation via site-specific, but diverse, mechanisms. These findings open the path to the rational design of new azurin mutants with different E0.

 Please wait while we load your content...
Please wait while we load your content...