Imidazoles from nitroallylic acetates and α-bromonitroalkenes with amidines: synthesis and trypanocidal activity studies†
Abstract
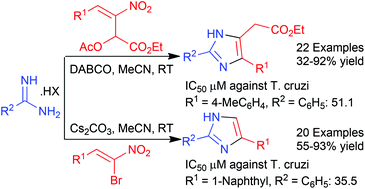
Cascade reactions of amidines with nitroallylic acetates and α-bromonitroalkenes provide potentially bioactive imidazoles in good to excellent yields in most cases. While 2,4-disubstituted imidazol-5-yl acetates are formed in the first case, 2,4-disubstituted imidazoles, bearing no substituent at position 5, are the products in the second case. These two series of imidazoles, viz. 2,4,5-trisubstituted and 2,4-disubstituted, were screened for their activity against the protozoan parasite Trypanosoma cruzi which is responsible for Chagas disease. As many as three compounds were as active as the standard benznidazole and two others were 2–3-fold more active highlighting the potential of substituted imidazoles, easily accessible from nitroalkenes, as possible anti-parasitic agents.

 Please wait while we load your content...
Please wait while we load your content...