One-pot synthesis of 1,3-enynes with a CF3 group on the terminal sp2 carbon by an oxidative Sonogashira cross-coupling reaction†
Abstract
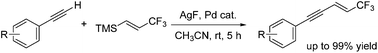
Oxidative Sonogashira cross-coupling reactions of (E)-trimethyl(3,3,3-trifluoroprop-1-enyl)silane with arylacetylene were achieved using silver fluoride and a palladium catalyst, to afford high yields of various 1,3-enynes with a CF3 group on the terminal sp2 carbon. Silver fluoride promoted C–Si bond dissociation and oxidation of palladium, enabling catalytic use of palladium.

 Please wait while we load your content...
Please wait while we load your content...