A potent and selective C-11 labeled PET tracer for imaging sphingosine-1-phosphate receptor 2 in the CNS demonstrates sexually dimorphic expression†
Abstract
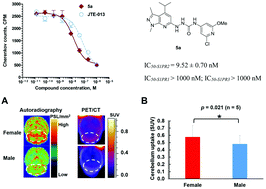
Sphingosine-1-phosphate receptor 2 (S1PR2) plays an essential role in regulating blood–brain barrier (BBB) function during demyelinating central nervous system (CNS) disease. Increased expression of S1PR2 occurs in disease-susceptible CNS regions of female versus male SJL mice and in female multiple sclerosis (MS) patients. Here we reported a novel sensitive and noninvasive method to quantitatively assess S1PR2 expression using a C-11 labeled positron emission tomography (PET) radioligand [11C]5a for in vivo imaging of S1PR2. Compound 5a exhibited promising binding potency with IC50 value of 9.52 ± 0.70 nM for S1PR2 and high selectivity over S1PR1 and S1PR3 (both IC50 > 1000 nM). [11C]5a was synthesized in ∼40 min with radiochemistry yield of 20 ± 5% (decayed to the end of bombardment (EOB), n > 10), specific activity of 222–370 GBq μmol−1 (decayed to EOB). The biodistribution study in female SJL mice showed the cerebellar uptake of radioactivity at 30 min of post-injection of [11C]5a was increased by Cyclosporin A (CsA) pretreatment (from 0.84 ± 0.04 ID% per g to 2.21 ± 0.21 ID% per g, n = 4, p < 0.01). MicroPET data revealed that naive female SJL mice exhibited higher cerebellar uptake compared with males following CsA pretreatment (standardized uptake values (SUV) 0.58 ± 0.16 vs. 0.48 ± 0.12 at 30 min of post-injection, n = 4, p < 0.05), which was consistent with the autoradiographic results. This data suggested that [11C]5a had the capability in assessing the sexual dimorphism of S1PR2 expression in the cerebellum of the SJL mice. The development of radioligands for S1PR2 to identify a clinical suitable S1PR2 PET radiotracer, may greatly contribute to investigating sex differences in S1PR2 expression that contribute to MS subtype and disease progression and it will be very useful for detecting MS in early state and differentiating MS with other patients with neuroinflammatory diseases, and monitoring the efficacy of treating diseases using S1PR2 antagonism.

 Please wait while we load your content...
Please wait while we load your content...