Synthesis of new carolacton derivatives and their activity against biofilms of oral bacteria†
Abstract
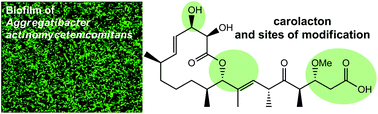
Carolacton, a secondary metabolite isolated from the extracts of Sorangium cellulosum, causes membrane damage and cell death in biofilms of the caries- and endocarditis-associated bacterium Streptococcus mutans. Here, we report the total synthesis of several derivatives of carolacton. All new structural modifications introduced abolished its biological activity, including subtle ones, such as inversion of configuration at C9. However, a bicyclic bislactone derivative as well as the methyl ester of carolacton resulted in compounds with prodrug properties. Their inhibitory activity on S. mutans was proven to be based on enzymatic hydrolysis by S. mutans which provided native carolacton resulting in biofilm damage in vivo. Moreover, we demonstrate that carolacton acts also on S. gordonii, S. oralis and the periodontitis pathogen Aggregatibacter actinomycetemcomitans, causing elongated cells and growth inhibition.

 Please wait while we load your content...
Please wait while we load your content...