Self-assembled polymeric nanocarriers for the targeted delivery of retinoic acid to the hair follicle†
Abstract
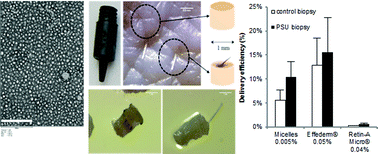
Acne vulgaris is a highly prevalent dermatological disease of the pilosebaceous unit (PSU). An inability to target drug delivery to the PSU results in poor treatment efficacy and the incidence of local side-effects. Cutaneous application of nanoparticulate systems is reported to induce preferential accumulation in appendageal structures. The aim of this work was to prepare stable polymeric micelles containing retinoic acid (RA) using a biodegradable and biocompatible diblock methoxy-poly(ethylene glycol)-poly(hexylsubstituted lactic acid) copolymer (MPEG-dihexPLA) and to evaluate their ability to deliver RA to skin. An innovative punch biopsy sample preparation method was developed to selectively quantify follicular delivery; the amounts of RA present were compared to those in bulk skin, (i.e. without PSU), which served as the control. RA was successfully incorporated into micelle nanocarriers and protected from photoisomerization by inclusion of Quinoline Yellow. Incorporation into the spherical, homogeneous and nanometer-scale micelles (dn < 20 nm) increased the aqueous solubility of RA by >400-fold. Drug delivery experiments in vitro showed that micelles were able to deliver RA to porcine and human skins more efficiently than Retin-A® Micro (0.04%), a marketed gel containing RA loaded microspheres, (7.1 ± 1.1% vs. 0.4 ± 0.1% and 7.5 ± 0.8% vs. 0.8 ± 0.1% of the applied dose, respectively). In contrast to a non-colloidal RA solution, Effederm® (0.05%), both the RA loaded MPEG-dihexPLA polymeric micelles (0.005%) and Retin-A® Micro (0.04%) displayed selectivity for delivery to the PSU with 2-fold higher delivery to PSU containing samples than to control samples. Moreover, the micelle formulation outperformed Retin-A® Micro in terms of delivery efficiency to PSU presenting human skin (10.4 ± 3.2% vs. 0.6 ± 0.2%, respectively). The results indicate that the polymeric micelle formulation enabled an increased and targeted delivery of RA to the PSU, potentially translating to a safer and more efficient clinical management of acne.

 Please wait while we load your content...
Please wait while we load your content...