Towards graphene iodide: iodination of graphite oxide†
Abstract
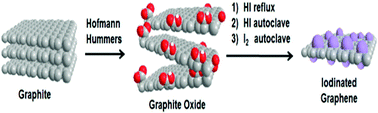
Halogenated graphene derivatives are interesting owing to their outstanding physical and chemical properties. In this paper, we present various methods for the synthesis of iodinated graphene derivatives by the iodination of graphite oxides prepared according to either the Hummers or Hofmann method. Both graphite oxides were iodinated by iodine or hydroiodic acid under reflux or in an autoclave at elevated temperatures (240 °C) and pressures (over 100 bar). The influence of both graphite oxide precursors on the properties of resulting iodinated graphenes was investigated by various techniques, including SEM, SEM-EDS, high-resolution XPS, FTIR, STA, and Raman spectroscopy. Electrical resistivity was measured by a standard four point technique. In addition, the electrochemical properties were investigated by cyclic voltammetry. Although the iodinated graphenes were structurally similar, they had remarkably different concentrations of iodine. The most highly iodinated graphenes (iodine concentration above 30 wt%) exhibited relatively high C/O ratios, confirming high degrees of reduction. Iodine is incorporated in the form of covalent bonds to carbon atoms or as polyiodide anions non-covalently bonded through the charge transfer reaction with the graphene framework. Iodinated graphenes with such properties could be used as the starting material for further chemical modifications or as flame-retardant additives.

 Please wait while we load your content...
Please wait while we load your content...