Studies on the reactions between the DNA bases and a model α,β-unsaturated oxoaldehyde†
Abstract
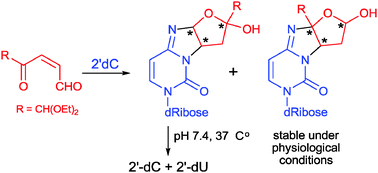
Structures of adducts formed in the reactions between a model oxoenal and the DNA bases were determined based on 2D NMR spectroscopy. Results obtained from quantum-mechanical studies indicated that two mechanisms can be involved in the generation of two pairs of stereomeric adducts of the oxoenal with 2′-deoxycytidine, and that the preference of one of the plausible routes depends on the structure of the forming product. Instability of one of these products leads to deamination of 2′-deoxycytidine. In the light of these results, stability of the structurally analogous 2′-deoxycytidine adducts of cis-2-butene-1,4-dial, the mutagenic metabolite of furan, was also studied, and it was demonstrated that these compounds undergo partial degradation resulting in the formation of 2′-deoxyuridine. This fact is of great importance with respect to biological consequences of the possible presence of such modifications in vivo. The data provide support to the hypothesis that in addition to 1,N6-etheno-2′-deoxyadenosine and 1,N2-etheno-2′-deoxyguanosine adducts of cis-2-butene-1,4-dial, also oxadiazabicyclooctaimine derivatives of 2′-deoxycytidine can significantly contribute to furan carcinogenic effects.

 Please wait while we load your content...
Please wait while we load your content...