Solvent effect and fluorescence response of the 7-tert-butylpyrene-dipicolylamine linkage for the selective and sensitive response toward Zn(ii) and Cd(ii) ions†
Abstract
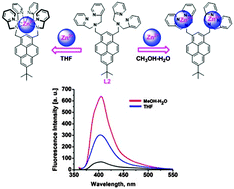
The different binding behaviour of 7-tert-butylpyrene based chemosensors bearing dipicolylamine (Dpa) linkages at the 1,3-positions was investigated in various solvents for the sensing of Zn(II) and Cd(II).The potential mono-chelating ligand L1 follows the same binding pattern in both THF and methanol–water solvent systems, exhibiting higher selectivity and sensitivity for Cd(II) over Zn(II) mainly in THF solvent system. The potential bis-chelate ligand L2 can selectively bind both Zn(II) and Cd(II) in a 1 : 1 ratio in THF, whereas in methanol–water (7 : 3) at pH = 7.0; a 1 : 2 binding ratio was observed. In THF, two sites of ligand L2 can only selectively and sensitively bind one Zn(II) or Cd(II). The different complexation behaviour of L1 and L2 in different solvents were studied by means of fluorescence spectra and 1H-NMR titration experiments in the presence of Zn(II) and Cd(II).


 Please wait while we load your content...
Please wait while we load your content...