The role of N-terminal proline in stabilizing the Ant–Pro zipper motif†
Abstract
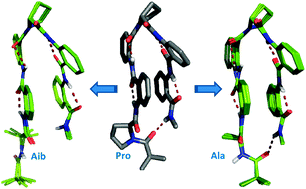
Hetero-chiral hybrid peptides of the general sequence LαβnDαβn featuring proline (Pro, a constrained α-amino acid) and anthranilic acid (Ant, a constrained β-amino acid) as building blocks, where n = 2, 4 etc., form a three-dimensional zipper-like architecture. These zipper peptides attain stable conformation by balancing the co-operative contribution of two competing non-covalent forces, namely hydrogen bonding and aromatic stacking. However, the selection of the N-terminal residue also stands to be one of the key contributors in stabilising the unusually long-range intramolecular hydrogen bond, featuring 26 atoms in the H-bonded ring observed at the termini. This article deals with the substitution alterations at the N-terminus of the zipper motif and their consequent influences on its structure and stability. In this study, the N-terminal Pro residue of the zipper motif was substituted with a flexible amino acid, alanine, and a constrained acyclic amino acid, 2-aminoisobutyric acid, to investigate the role of N-terminal proline in stabilizing the Ant–Pro zipper motif, and its stabilities were assessed by employing solution-state NMR and restrained MD simulation studies.
- This article is part of the themed collection: Foldamer Chemistry

 Please wait while we load your content...
Please wait while we load your content...