New thermally stable energetic materials: synthesis and characterization of guanylhydrazone substituted furoxan energetic derivatives†
Abstract
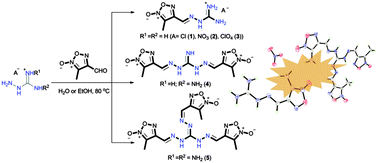
New guanylhydrazone substituted furoxan energetic derivatives were synthesized via the condensation reactions of 3-methyl-4-furoxancarbaldehyde with aminoguanidine derivatives. The resulting compounds 1–5 were well characterized using IR spectroscopy, multinuclear NMR spectroscopy, differential scanning calorimetry, thermogravimetry analysis as well as elemental analysis. Additionally, the structures of compounds 1, 2 and 5 were confirmed by single crystal X-ray diffraction. Except for the compounds 1 and 2, all the remaining products exhibit good thermal stabilities with decomposition temperatures above 200 °C. The detonation pressure values calculated for these compounds range from 17.0 to 28.3 GPa, and the detonation velocities range from 6906 to 8210 m s−1. These values suggest that the guanylhydrazone substituted furoxan energetic derivatives could be potential candidates for thermally stable energetic materials.

 Please wait while we load your content...
Please wait while we load your content...