Analyzing free zinc(ii) ion concentrations in cell biology with fluorescent chelating molecules
Abstract
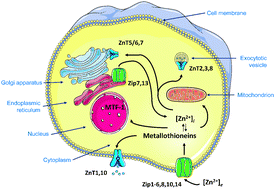
Essential metal ions are tightly controlled in biological systems. An understanding of metal metabolism and homeostasis is being developed from quantitative information of the sizes, concentrations, and dynamics of cellular and subcellular metal ion pools. In the case of human zinc metabolism, minimally 24 proteins of two zinc transporter families and a dozen metallothioneins participate in cellular uptake, extrusion, and re-distribution among cellular compartments. Significantly, zinc(II) ions are now considered signaling ions in intra- and intercellular communication. Such functions require transients of free zinc ions. It is experimentally quite challenging to distinguish zinc that is protein-bound from zinc that is not bound to proteins. Measurement of total zinc is relatively straightforward with analytical techniques such as atomic absorption/emission spectroscopy or inductively coupled plasma mass spectrometry. Total zinc concentrations of human cells are 200–300 μM. In contrast, the pool of non-protein bound zinc is mostly examined with fluorescence microscopy/spectroscopy. There are two widely applied fluorescence approaches, one employing low molecular weight chelating agents (“probes”) and the other metal-binding proteins (“sensors”). The protein sensors, such as the CALWY, Zap/ZifCY, and carbonic anhydrase-based sensors, can be genetically encoded and have certain advantages in terms of controlling intracellular concentration, localization, and calibration. When employed correctly, both probes and sensors can establish qualitative differences in free zinc ion concentrations. However, when quantitative information is sought, the assumptions underlying the applications of probes and sensors must be carefully examined and even then measured pools of free zinc ions remain methodologically defined. A consensus is building that the steady-state free zinc ion concentrations in the cytosol are in the picomolar range but there is no consensus on their concentrations in subcellular compartments. Applying the extensive toolbox of available probes/sensors in biological systems requires an understanding of the principles of cellular zinc homeostasis and the chemical biology of the probes and sensors. Regardless of limitations in specificity (for a particular metal ion), selectivity (for a particular metal pool), and sensitivity (detection limit), the technology is making remarkable contributions to imaging zinc with high spatiotemporal resolution in single cells and to defining the biochemical functions of zinc ions in cellular regulation.
- This article is part of the themed collections: Zinc in the Biosciences and Iron and zinc sensing in cells and the body

 Please wait while we load your content...
Please wait while we load your content...