Comparative binding mechanism of lupeol compounds with plasma proteins and its pharmacological importance†
Abstract
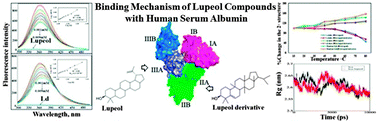
Lupeol, a triterpene, possesses beneficial effects like anti-inflammatory and anti-cancer properties. Binding of lupeol and its derivative (phytochemicals) to plasma proteins such as human serum albumin (HSA) and α-1-acid glycoprotein (AGP) is a major determinant in the disposition of drugs. Cytotoxic studies with mouse macrophages (RAW 246.7) and HeLa cell lines revealed anti-inflammatory and anti-cancer properties for both lupeol and lupeol derivative. Both molecules reduced the expression of pro-inflammatory cytokines in LPS induced macrophages. Further, apoptosis was observed in HeLa cell lines when they were incubated with these molecules for 24 h. The fluorescence quenching of HSA was observed upon titration with different concentrations of lupeol and lupeol derivative; their binding constants were found to be 3 ± 0.01 × 104 M−1 and 6.2 ± 0.02 × 104 M−1, with binding free energies of −6.59 kcal M−1 and −7.2 kcal M−1. With AGP, however, the lupeol and lupeol derivative showed binding constants of 0.9 ± 0.02 × 103 M−1 and 2.7 ± 0.01 × 103 M−1, with free energies of −4.6 kcal M−1 and −5.1 kcal M−1 respectively. Molecular displacement studies based on competition with site I-binding phenylbutazone (which binds site I of HSA) and ibuprofen (which binds site II) suggest that lupeol binds site II and the lupeol derivative site I. Molecular docking studies also confirmed that lupeol binds to the IIIA and the lupeol derivative to the IIA domain of HSA. Secondary structure changes were observed upon formation of HSA–lupeol/lupeol derivative complexes by circular dichroism spectroscopy. Molecular dynamics simulations support greater stability of HSA–lupeol and HSA–lupeol derivative complexes compared to that of HSA alone.
- This article is part of the themed collection: Chemical Biology in Molecular BioSystems

 Please wait while we load your content...
Please wait while we load your content...