Structural and dynamic basis of acid amido synthetase GH3.1: an investigation of substrate selectivity and major active site access channels†
Abstract
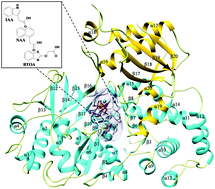
Auxin/IAA (Indole-3-acetic acid) plays critical roles in many aspects of plant growth and development. Gretchen Hagen 3.1 (GH3.1) enzyme from grapevine (Vitis vinifera) catalyzes the ATP-dependent conjugation of aspartate to IAA suggesting a significant way to modulate levels of cellular auxins/IAA. It is reported that VvGH3.1 prefers IAA as the substrate than 1-naphthaleneacetic acid (NAA) and benzothiazole-2-oxyacetic acid (BTOA), whereas the detailed interaction mechanism of these substrates remains unclear. In this study, based on the recently reported crystal structure of VvGH3.1 and AtGH3.12, the open form of VvGH3.1 was built. Then combined computational techniques including molecular docking, molecular dynamics (MD) simulations, Molecular Mechanics Generalized Born Surface Area and Poisson–Boltzmann Surface Area (MM-GB/PBSA) methods and active site access channel analysis were utilized to investigate the binding mechanism of three substrates, IAA, NAA and BTOA, and dynamic behaviors of GH3.1 induced by substrate binding. The predicted binding free energy is in agreement with the experimental work with an order of IAA < NAA < BTOA. Key residues interacting with three substrates and residues specifically binding to one of them were identified. The strong hydrogen bond interaction formed between IAA and Ser108, Ser339, Gln560 makes it the best substrate over NAA and BTOA. The dynamic behaviors, especially in the dominant access channels of IAA and NAA bound GH3.1, were found to be different from those in the BTOA–GH3.1 system with larger bottleneck radius and shorter length. This study provided novel insight to understand the substrate selectivity mechanism of GH3.1 and also for the rational design of novel auxin-based herbicides and growth regulators.

 Please wait while we load your content...
Please wait while we load your content...