Synchrotron-based X-ray spectromicroscopy and electron paramagnetic resonance spectroscopy to investigate the redox properties of lead chromate pigments under the effect of visible light†
Abstract
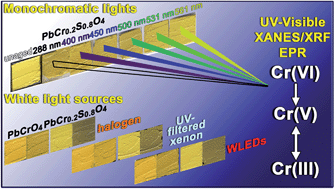
Light-induced redox processes have been established as the cause of the chromatic alterations of a number of artists' pigments used from the 15th to the 20th century. Despite the fact that a general comprehension of the mechanisms has been provided through the characterization of photo-degraded compounds, both exhaustive information on the wavelength-dependence of the alteration process of the pigments and experimental evidence in how visible light may influence the formation pathways of specific secondary compounds are still lacking. Establishing an analytical protocol for the study of wavelength-dependence of pigments on photo-redox pathways is relevant for the safe illumination of paintings, especially in view of the possible use of spectrally tunable light sources such as white light emitting diodes (WLEDs). In this work, we propose an integrated approach based on a combination of diffuse reflectance UV-visible, synchrotron radiation (SR)-based micro X-ray fluorescence (μ-XRF)/X-ray absorption near edge structure (μ-XANES) and electron paramagnetic resonance (EPR) spectroscopies to study the photo-redox process of Cr(VI) → Cr(III) for lead chromate yellows (PbCr1−xSxO4, 0 ≤ x ≤ 0.8) under exposure to different monochromatic light. In view of the thin (3–5 μm) alteration layer that is formed at the paint surface after light exposure, SR-based Cr K-edge μ-XANES/μ-XRF analysis was employed to obtain information on the abundance, nature and distribution of the alteration of Cr(III)-compounds at the micrometric-scale level. On the other hand, EPR spectroscopy was used as a complementary tool to the SR-based X-ray methods due to its sensitivity for revealing species containing one or more unpaired electrons and for distinguishing different coordination geometries of paramagnetic centers, such as Cr(V)-species. Semi-quantitative indications about the darkening of the paint surface were obtained by UV-Vis spectroscopy. An abundance of reduced Cr down to around 50% was detected at the aged surface of chrome yellow paints. The reduction process was favored not only by wavelengths shorter than 460 nm (i.e., where the pigment shows its maximum absorption) but also by light in the 490–530 nm range. The first evidence of the presence of Cr(V)-intermediates in the Cr(VI) → Cr(III) reduction reaction allowed the risks of inducing photo-degradation of the 490–530 nm wavelength range to be explained.

 Please wait while we load your content...
Please wait while we load your content...