Synthesis of glucose-mediated Ag–γ-Fe2O3 multifunctional nanocomposites in aqueous medium – a kinetic analysis of their catalytic activity for 4-nitrophenol reduction†
Abstract
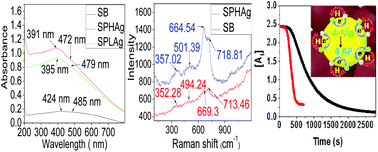
This paper reports the synthesis of γ-Fe2O3 supported Ag nanoparticles (NPs) in aqueous medium by following a green approach. The presence of Fe2O3 in the gamma phase and silver in the nanocomposite has been confirmed by Raman spectroscopy, EDAX and XPS analyses. The presence of Ag in the nanocomposite is also indicated by UV spectroscopy. In the process of in situ generation of glucose mediated Ag NPs on the γ-Fe2O3 matrix, the size of γ-Fe2O3 nanoclusters reduced from 11.6 ± 1.6 to 9 ± 1 nm as was estimated from HRTEM analysis. Glucose served as an effective stabilizer for both Ag and γ-Fe2O3 in the nanocomposite. At lower concentrations of Ag (0.15–1.2 μM) the reduction of 4-nitrophenol (4-Nip) follows pseudo-first-order kinetics and the second order rate constant for this process was found to be 5.28 × 103 dm3 mol−1 s−1. Whereas, at higher concentrations (3.2–28.9 μM), it follows zero-order kinetics and occurred with a rate constant of 1 × 10−2 mol dm−3 s−1. The amount of silver in the nanocomposite is found to influence the kinetics of the catalytic reduction in a complex scheme following the Langmuir–Hinshelwood mechanism. The recyclability of the as-synthesized nanocomposite up to 7 cycles and the catalytic effect even at a very low silver concentration (0.15 μM) associated with high surface area and superparamagnetism suggest it to be a cost effective and environmentally friendly potential catalytic system.

 Please wait while we load your content...
Please wait while we load your content...