Effects of the MoO3 structure of Mo–Sn catalysts on dimethyl ether oxidation to methyl formate under mild conditions†
Abstract
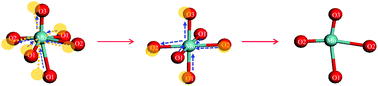
The selective oxidation of dimethyl ether (DME) to methyl formate (MF) was conducted in a fixed-bed reactor over the MoO3–SnO2 catalysts with different Mo/Sn ratios. The MF selectivity reached 94.1% and the DME conversion was 33.9% without the formation of COx over the MoSn catalyst at 433 K. The catalysts were deeply characterized by NH3-TPD, CO2-TPD, BET, XPS and H2-TPR. The characterization results showed that different compositions of catalysts obviously affected the surface properties of the catalysts, but the valence of the metal hardly changed with the Mo/Sn ratios. Raman spectroscopy, XRD and XAFS were further used to characterize the structure of the catalysts. The results indicated that the catalyst composition exerted a significant influence on the structure of MoO3. The formation of oligomeric MoO3 and the appropriate coordination numbers of Mo–O at 1.94 Å are the main reasons for the distinct high catalytic activity of the MoSn catalyst.

 Please wait while we load your content...
Please wait while we load your content...