A low content Au-based catalyst for hydrochlorination of C2H2 and its industrial scale-up for future PVC processes†
Abstract
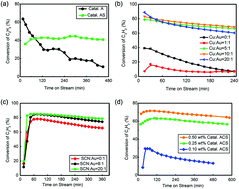
China has the world's largest polyvinyl chloride (PVC) production capacity, comprising over 20 Mt a−1 and occupying 41% of the world production capacity. However, the production of the PVC monomer, vinyl chloride monomer (VCM), faces unsustainable development due to mercury problems. Over 70% of VCM in mainland China is synthesized through hydrochlorination of C2H2 catalyzed by HgCl2. Mercury and its compounds escaping from the reactors have high chronic toxicity, which is harmful to the environment and to people's health. Therefore, developing a novel mercury-free catalyst is crucial for maintaining a green production of PVC in China. This paper shows a novel low content Au-based catalyst by complexing Au with thiocyanate (–SCN). This chemical complex significantly decreases the electrode potential of Au3+ from 0.926 V to 0.662 V, and hence reduces the probability of its reduction by C2H2. The catalyst preserves a high turnover frequency of 5.9 s−1 based on Au, and over 3000 h testing of a 4 t a−1 pilot-trial shows its promising reactivity (>95%) and selectivity (>99%). Compared with the conventional HgCl2 catalyst, this novel Au catalyst has better reactivity, stability, environmental friendliness and lower toxicity, making it promising for the sustainable development of China's PVC industry.

 Please wait while we load your content...
Please wait while we load your content...