Environmental photochemistry of single layered graphene oxide in water†
Abstract
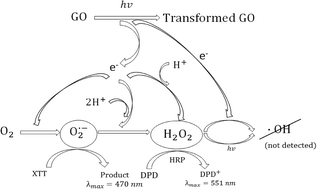
Graphene oxide (GO) is a carbonaceous nanomaterial that is a precursor material in the preparation of graphene, and because of its unique properties, it will likely be used in a number of industrial and consumer products in the future. Despite its name, it contains many epoxy, hydroxyl, and carboxyl functional groups on its edges and surface, making it easy to suspend in water. However, how it is transformed or mineralized in natural aquatic environments and its effects on natural processes within these environments remain largely unknown. Therefore, in this study, we report on the photochemical reactivity of single layered GO dispersed in water and irradiated with light within the solar spectrum that reaches the water bodies at the earth's surface (λ ≥ 300 nm). Upon irradiation, the visible color of a 5 mg L−1 GO suspension shifted from pale to dark brown, possibly indicating the repair of some of the π-bond structures; however, Raman spectroscopy indicated an increase in nonaromatic defects. To further examine how oxidation or reduction of the GO surface may occur upon solar light irradiation, we probed the production of various reactive oxygen species (ROS). By monitoring ROS production with selective and highly reactive chemical probes, formation of superoxide anions (O2˙−), but not single oxygen (1O2) or hydroxyl radicals (·OH), was detected, indicating electron transfer from GO to dissolved molecular oxygen (O2). However, further electron transfer through reduction of O2˙− did occur, as hydrogen peroxide (H2O2) was found to accumulate, forming 3 μM H2O2 in a suspension of 5 mg L−1 GO after 4 hours of irradiation.

 Please wait while we load your content...
Please wait while we load your content...