Scalable synthesis of bi-functional high-performance carbon nanotube sponge catalysts and electrodes with optimum C–N–Fe coordination for oxygen reduction reaction†
Abstract
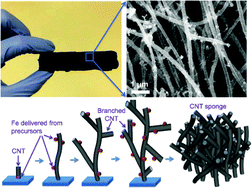
Oxygen reduction reaction (ORR) is essential in various electrochemical energy conversion processes, but its sluggish kinetics calls for catalysts made of platinum or its alloys. Although their high catalytic activity has been hardly challenged, the high price of the precious metal has limited their wide applications. Nitrogen-doped carbonaceous materials have been reported as alternatives due to the low cost of carbon and nitrogen precursors, but the low-cost has accompanied by low catalytic activity and poor stability, particularly in acidic media. Here we developed 3-dimentional (3D) N/Fe-containing carbon nanotube (CNT) sponges showing striking improvements in catalytic activity and stability in both acidic and basic solutions. The onset potential and limiting current density in 0.5 M H2SO4 or 0.1 M KOH were comparable to those of Pt/C (20 wt% Pt). More importantly, cyclic voltammetry (CV) tests up to 30 000 cycles suggest their excellent long-term stability even better than those of Pt/C. We believe that the key for the high performance is pyridinic nitrogen coordinated with iron in 3D CNTs, according to the comparative studies with their variants whose characteristics include iron-deficiency as well as nitrogen doping with weak iron coordination. This study demonstrates that a proper doping, coordination, and morphology design of CNT bulks could lead to an outstanding performance, and the findings will be of great value for further improving non-precious metal catalysts. The self-standing and porous sponge-like 3D structure could substantially facilitate the mass transfer for ORR and, therefore, potentially act as a gas diffusion layer in electrochemical cells. The unique bi-functionality with a low cost but a high performance could make fuel cells as commercially viable options in the future.


 Please wait while we load your content...
Please wait while we load your content...