Group 13 complexes of dipyridylmethane, a forgotten ligand in coordination chemistry†
Abstract
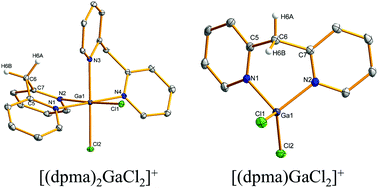
The reactions of dipyridylmethane (dpma) with group 13 trichlorides were investigated in 1 : 1 and 1 : 2 molar ratios using NMR spectroscopy and X-ray crystallography. With 1 : 1 stoichiometry and Et2O as solvent, reactions employing AlCl3 or GaCl3 gave mixtures of products with the salt [(dpma)2MCl2]+[MCl4]− (M = Al, Ga) as the main species. The corresponding reactions in 1 : 2 molar ratio gave similar mixtures but with [(dpma)MCl2]+[MCl4]− as the primary product. Pure salts [(dpma)AlCl2]+[Cl]− and [(dpma)AlCl2]+[AlCl4]− could be obtained by performing the reactions in CH3CN. In the case of InCl3, a neutral monoadduct (dpma)InCl3 formed regardless of the stoichiometry employed. A neutral adduct (dpma)(BCl3)2 was obtained from the reaction between dpma and BCl3 in Et2O using 1 : 2 stoichiometry. With 1 : 1 molar ratio of reagents, a mixture of products and deprotonation of the methylene bridge in [(dpma)BCl2]+ was observed. The experimental data showed that the structural flexibility of the dpma ligand results in more diverse coordination chemistry with group 13 elements than that observed for bipyridine (bpy), while computational investigations indicated that the investigated metal–ligand interactions are, to a first approximation, independent of the ligand type. Electrochemical and chemical attempts to reduce the cations [(dpma)MCl2]+ showed that, in stark contrast to the chemistry of the related [(bpy)BCl2]+ cation, the neutral radicals [(dpma)MCl2]˙ are extremely unstable. Differences in the redox behaviour of dpma and bpy could be rationalized with the electronic structure of the ligand and that of the methylene bridge in particular. As a whole, the facile reactivity of the methylene bridge in the dpma ligand renders it amenable to further reactivity and functionalization that is not possible in the case of bpy.

 Please wait while we load your content...
Please wait while we load your content...